Correct Answer

verified
Correct Answer
verified
Essay
The elements known as the halogens are useful as disinfectants. Name two halogens.
Correct Answer

verified
(two of th...View Answer
Show Answer
Correct Answer
verified
View Answer
Essay
Describe the contributions of Marie Curie.
Correct Answer

verified
(note that answers will vary)Marie Curie...View Answer
Show Answer
Correct Answer
verified
View Answer
True/False
Marie Curie suggested the name "radioactivity" to describe the spontaneous emission of particles and/or radiation.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Give the number of protons (p) , electrons (e) , and neutrons (n) in one atom of nickel-62.
A) 28 p, 28 e, 28 n
B) 28 p, 28 e, 34 n
C) 28 p, 28 e, 62 n
D) 62 p, 28 e, 28 n
E) 62 p, 62 e, 28 n
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An aluminum ion, Al3+, has:
A) 13 protons and 13 electrons
B) 27 protons and 24 electrons
C) 16 protons and 13 electrons
D) 13 protons and 10 electrons
E) 10 protons and 13 electrons
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Atoms of the same element with different mass numbers are called
A) ions.
B) neutrons.
C) allotropes.
D) chemical families.
E) isotopes.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mineral pyrolusite is a compound of manganese-55 and oxygen-16. If 63% of the mass of pyrolusite is due to manganese, what is the empirical formula of pyrolusite?
A) MnO
B) Mn2O
C) Mn2O2
D) MnO2
E) none of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
Give the formula of magnesium nitrate.
Correct Answer

verified
Correct Answer
verified
Short Answer
Use the following to answer questions 70-76: 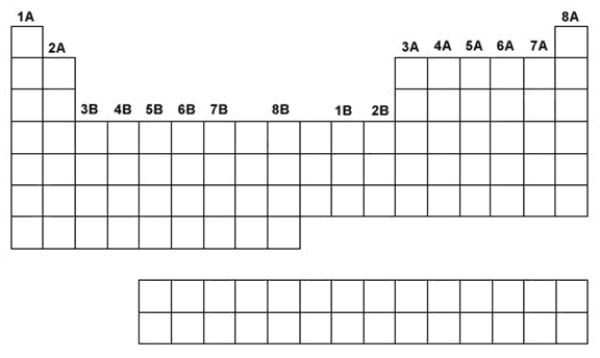 -Use the periodic table above to show where the alkali metals are located.
-Use the periodic table above to show where the alkali metals are located.
Correct Answer

verified
Correct Answer
verified
Short Answer
Write the formula for the acid formed from the hydrogen sulfate anion, and then name the acid.
Correct Answer

verified
Correct Answer
verified
Short Answer
Predict the formula for the binary compound formed between aluminum and fluorine.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Stock system name for Co2(SO3) 3 is:
A) cobalt sulfate
B) cobalt(II) sulfite
C) cobalt(II) sulfate
D) cobalt(III) sulfite
E) cobalt(III) sulfate
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
How many protons are there in one atom of nickel?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following elements is most likely to form a 2- ion?
A) scandium
B) selenium
C) silicon
D) strontium
E) iodine
G) None of the above
Correct Answer

verified
Correct Answer
verified
Short Answer
How many electrons, protons, and neutrons are in a neutral atom of the following isotope of gadolinium? 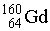
Correct Answer

verified
64 electro...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
When J. J. Thomson discovered the electron, what physical property of the electron did he measure?
A) its charge, e
B) its charge-to-mass ratio, e/m
C) its temperature, T
D) its mass, m
E) its atomic number, Z
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The Stock system name for As2S5 is
A) arsenic(V) sulfide.
B) diarsenic pentasulfide.
C) arsenic(III) sulfide.
D) arsenic(V) sulfate.
E) diarsenic sulfate.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which one of the following elements is most likely to form a 2+ ion?
A) beryllium
B) carbon
C) fluorine
D) oxygen
E) sodium
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
What are the seven elements that naturally occur as diatomic molecules?
Correct Answer

verified
Hydrogen, nitrogen, ...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 61 - 80 of 141
Related Exams